did you know microwave radiation only (?) heats substances with polar molecules. As they rotate and generate heat through molecular friction
Tollens reagent
used to detect aldehydes. It has a formula of [Ag(NH3)2].
SN1 and SN2 reactions
background: a chemical reaction rarely occurs in one step, this would involve a great deal of energy and the chance of particles all coliding with the correct orientation all at the same time is unlikely, hence , chemical reactions occur via a series of steps , each step has its own rate of reaction, one step will be the slowest step. this affects the overall rate of the entire reaction. this step is called the slowest step or rate determining step. particles which take palace in this step will effect the overall reaction and its rate. in an SN1 reaction The leaving group detached first before the incoming nucleophile attaches , hence the rate depends only on how quickly the leaving group detaches and it is the leaving group which is in the rate determining step.
However, in an SN2 reaction , as the leaving group detaches, so the incoming group gets attached simoultaneously, forming an extremely unstable high energy transition state for a brief moment, in this case the rate of the reaction is dependent on both how quickly the leaving group leaves and how quickly the incoming group joins. Both leaving group and incoming nucleophilc are part of rate determining step.
To determine wether a reaction is SN1 or SN2 we must carry out a series of experiments where we change the conc of the reactant particles one at a time and note how each change affects the overall rate of the reaction. This will help us determine the reactants which take part in the rate determining step and given that, we can deduce if the reaction is Sn1 or Sn2. This experimental analysis will also enable us to form a rate equation for the reaction
Benzene
Benzene is an organic molecule which is formed of 6 cyclicaly bonded carbons which share some delocalised electrons. Each carbon is also bonded to its very own hydrogen.
August Kekule thought that benzene was composed of alternatngly double bonded carbons. But he was completely wrong
Equilibrium
Le Chatelier's principle
Magic rule for equilibrium reactions: basically everything you change in the system it's going to hate. This where the classic saying "there is a similarity between conservative beleifs and equilibrium reactions, that similarity is that they are both opposed to change" comes from
If you increase the presure the system will favour the side with the fewest moles, decreasing the presure
If you increase the temperature the system will favour the enothermic reaction, decreasing the temperature
If you increase the concentration/ammount of any of the species, the oposing species will be more readily produced
And vice versa for all of these.
I'm still not sure wether this is named after the title or the name of the guy that discovered this. #Update#, it's his last name. Henry Louis Le Chatelier

Esters
RCOOR functional group
esters are produced in a process called esterification (what a coincidence). An alcohol (OH) reacts with a caerboxilic acid (COOH). This reaction is reversible
Carboxylic acid + Alcohol <--> Ester + Water.
I would like to add more but i wont for vague reasons
They are also produced in an alternative way, which confusingly isn't called esterification.
This reaction isn't reversible.
An Acyl chloride plus an alcohol
Website Recommendations
MolView
Very easy to access and use, intuitive. Big fan of the 3D interface . My one quible is that you will need the premium version to draw any arrows (electron transfer, reversible and irriversible reaction) which is just really silly. Getting the arrows only requires exporting the file or even just screenshotting and dawing them on but its just an unnecesary faff, especially if you dont have a drawing pad (which I dont).
Friedel krafts acetylation
Acylation is when an acyl group is inserted into another molecule. Acyl groups being hydrocarbons attached to a carbon oxygen double bond, the carbon having another R group. Freidel-crafts acetylation has the addons of involving benzene and specifically acyl chlorides (Which just means that that R group I mentioned earlier is Chlorine)
The catalyst is aluminium chloride (AlCl3)
This is very useful for attaching hydrocarbon based groups to a benzene ring, the immediate product is a ketone, but if you already know how to mangle ketones into other things that’s no trouble.
Atomic structure and the periodic table summary sheet
Ionisation energy- first and second
first ionisation energy- the energy required to form 1 mole of +1 cations from one mole of an element
H(g) —-> H+(g) + e-
second ionisation energy- energy required for +1 cations to form 1 mole of gaseous +2 cations
H+(g) —-> H+2(g) + e-
factors that affect ionisation energy:
electrostatic force of attraction of the nucleus
distance of outer electrons from nucleus
electron shielding
note- ionisation energies will get larger and larger as more electrons are stripped off, this is because the more ionised the ion is, the greater the force of attraction from the nucleus on the remaining electrons
note - ionisation energies can be used to determine which element you’re observing. if there is a major drop you will know when the ion moves onto a lower shell
electronic structure
SPDF
S - holds 2 electrons
P - holds 6 electrons
D - holds 10 electrons
F - holds 14 electrons
all of which are split up into orbitals which contain 2 electrons of opposite spin
orbital shapes are estimations of likely places where you will find the electrons- its really more of a probability field
hydrogen, 1S1
helium, 1S2
lithium, 1S22S1
berryilum, 1S22S2
boron, 1S22S22P1
Amines, amides, amino acids and protein (basically the nitrogen containing organic compounds)
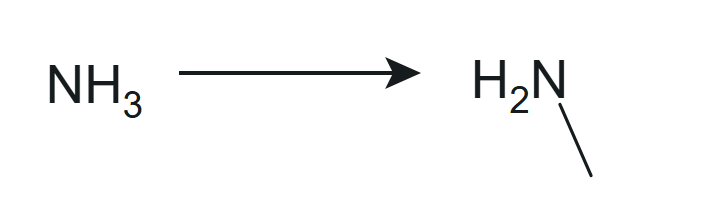
Amine - ammonia molecule with one of the hydrogens replaced by an alkyl or aryl group. If only one of the hydrogens is replaced, you will get a primary amine
Redox
Oxidation is the gain of electrons
Cl2 + 2e- —> 2Cl-
Buffer solutions
A buffer solution is a system that minimises pH changes when small amounts of an acid or base.
Buffer solution can be formed by 2 different methods
method 1: from a weak acid ands its salt ( eg CH3COOH and CH3COONA )
method 2: Via a partial neutralisation of a weak acid with a strong base ( eg CH3COOH and NaOH
CH3COOH ⇌ CH3COO- + H+
On addition of an acid, [H+] increases. These H+ ions react witht the conjugate base, the CH3COO-ion, causing the equilibrium position to shift to the left, removing most of the H+ ions.
On addition of an alkali, [OH-] increases. The small concentration of H+ ions react with these OH- ions to produce water. This causes more CH3COOH to disassociate and the equilibrium position to shift to the right, restoring the [H+].
Acid Base Equilibria
Bronsted-Lowry
A Bronsted-Lowry acid is defined as a substance that can donate a proton.
A Bronsted-Lowry base is defined as a substance that can (you’ll never guess…) accept a proton.
Calculating pH
Magic equation: pH + -log[H+]
Rearranging we can also get an even more magic equation for [H+] ...
[H+] = 10-pH
First ionisation energy down group 2 metals
The outermost electrons become further from the nucleus and that means weaker electrostatic attraction. which means , hoorayy, lower ionisation energy.
Furthermore you also get more electron shielding, which also lowers ionisation energy.
Poem
I once frolicked in my childhood field of geraniums
with my faithful companion, my lump of magnesium
when all of a sudden do I look at trotters, sodden
with dripping electrons all over my crocket and jone’s , bought at a premium
to which I am warned I have not brought my dutiful champion, my beloved magnesium
but rather some petulant barium
magnesium so gentle and trusting, keeps few friends but extremely closely
however barium that old clot, keeps plenty in frenzy
and cares not if some fall on my toesies
more to be said of barium that tool,
shields so many friends away from himself the old fool